

Timothy C. Edgell
University of New Brunswick, Saint John, Canada.
Europeans have sailed to North America for centuries. Not without consequence. Of perpetual concern-here in Canada and elsewhere-is the consequence of accidental species introductions.
As the trans-Atlantic shipping industry mushroomed, species exchanges between distant shores became inevitable. Early in the 1800s, European green crabs-Carcinus maenas-were transported to New England. Soon after, the crab's new world range expanded. They were first spotted north of Cape Cod in 1910. By 1950, green crabs were throughout the Gulf of Maine and on the verge of the Canadian Maritimes. In 1955, Fisheries Canada warned, "Watch for green crab - A new clam enemy".
Indeed, more than softshell clams were at risk. For example, the intertidal snail, Littorina obtusata, had an unfortunately thin shell before green crabs arrived. As quickly as 15 years post-invasion, these same snails developed thick-walled, crab-resistant shells. My colleagues and I, namely Geoff Trussell at Northeastern University and Brian Lynch, my former bench-mate, at the University of New Brunswick, set out to understand these changes. Knowing that snail behaviour and shell form were highly plastic traits, we aimed to test if developmental plasticity had evolved in L. obtusata. We did this by rearing British and American snails in the presence and absence of predator risk cues. As suspected, results showed a relation between plasticity variation and regional predation history.
Theory predicts that inducible defences canalize when predation risk is predictable, and evolve lability when predation risk is stochastic. Consistent with predictions, British snails, having co-existed with green crabs for centuries, had more canalized development than American conspecifics, having co-existed with green crabs for relatively less time-about 60 to 110 year, respectively-in the north and south Gulf of Maine, USA.
BOX 1:Inducible Behaviour After 35 days exposure to predator risk cues, snails from all populations exhibited adaptive behaviours. Some snails stopped feeding-body growth decelerated-presumably an attempt to avoid detection. Other snails kept feeding-body growth similar to controls-but enhanced antipredator defence (i.e., increased sensitivity to handling, increased soft-tissue retraction depth) presumably in anticipation of predation. Significantly, behaviours differed among regions. In the northern Gulf of Maine, defence behaviours were mixed. In the southern Gulf, some specialization appeared to emerge. Compare that to British snails, where populations always used one or the other defence behaviour, but never both simultaneously. Consistent with theory, canalization of behaviour correlated positively with predation history among regions. |
| BOX
2:Inducible Morphology.
After four months, risk cues induced adaptive changes in individual shell form-from thinner to thicker shell walls. Like behaviour, the magnitude of inducible change related to predation history: British snails were least plastic, American snails were most. Even within America, snails from a more recently-invaded region-northern Gulf-had greater plasticity than snails with a longer history of predation-southern Gulf. |
Given the pervasiveness of adaptive phenotypic plasticity in Molluscs and other phyla, results are likely applicable to other systems where species-species interactions vary in space or time. I (TCE) am currently posted at the Bamfield Marine Sciences Centre, British Columbia, Canada where green crabs invaded no more than ten years ago. As part of my current research programme, I study the green crab footprint on native Molluscs, in particular, predator recognition and the evolution of inducible defence in a common whelk, Nucella lamellosa.
I'd like to extend warm thanks to John W. Grahame, University of Leeds, UK for guidance and logistical support in Britain. Also to Geoff C. Trussell at Northeastern University Marine Science Center, Nahant, USA and Rémy Rochette at the University of New Brunswick, Saint John, Canada for provision of lab space. Finally, thanks to the Malacological Society of London for awarding initial funds.
Further Reading.
Edgell, T. C. and R. Rochette. 2007. Evol. Ecol. Res. 9: 579-597.
Medcof, J. C. and L. M. Dickie. 1955. Fish. Res. Canada Gen. Circ. No. 26.
Trussell, G. C. and L. D. Smith. 2000. Proc. Natl. Acad. Sci. USA. 97: 2123-2127
Fig. 1. The geography of Littorina obtusata.
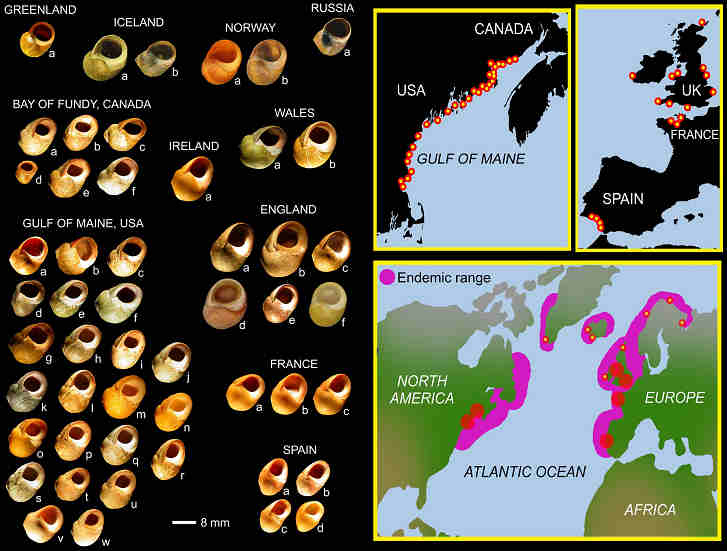
Fig. 2. Juvenile Littorina obtusata, tagged for later identification in a predation risk experiment.
