

Sam Stanton
Institute of Marine Sciences, School of Biological Sciences, University
of Portsmouth, Ferry Road, Portsmouth P04 9LY, UK
A combination of EM and fluorescence microscopy is being used to reveal the development of ciliation on the larval mantle and the velum of Crassostrea gigas larvae. These observations are providing insights into the functional importance of cilia-bearing mantle sense organs in, for example, settlement behaviour and of the velar cells in feeding mechanisms.
The catecholamines
Catecholamines are a group of neurotransmitters which have previously
been located within the larvae of Crassostrea gigas (Croll et
al. 1997; Croll and Dickinson 2004). They have been found to act as
morphogens in early development, as controllers of ciliary activity
and as inducers of settlement in the larva. Previous studies have used
methods such as liquid chromatography, or exposure of larvae to varying
catecholamine concentrations to demonstrate catecholamine presence and/or
effect. In the current study, the distribution of catecholamines has
been investigated using aldehyde-induced fluorescence. This information
is then compared with images from bright field microscopy and scanning
electron microscopy (SEM) to determine where catecholamines occur within
the larval anatomy.
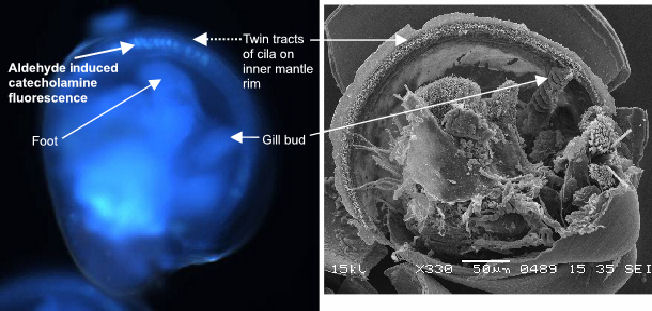
Catecholamine-positive cells have been found around the ventral rim
of the mantle and near the gill bud in larvae that are ready to settle
(pediveligers). The mantle signal occurs within the twin tracts of cilia
previously identified using the SEM. Figure
1 illustrates discrete patches of staining, perhaps individual cells,
whilst Figure 2
shows the same region using SEM.
 |
|
| Figure 1 UV image of catecholamine fluorescence in C.gigas pediveliger larva. | Figure 2 SEM of C.gigas pediveliger showing cilia on mantle rim and gill bud |
The gill bud appears as the point of origin for the inner mantle rim
ciliation in the veliger. As the larva develops from free-swimming veliger
to the swimming-crawling pediveliger ciliation increases. By late-stage
pediveliger mantle ciliation is dense in the region of the gill bud/mantle
rim join. It is especially interesting to note the presence of catecholamines
in the ventral region adjacent to the foot exiting between the valves
(Figure 1).
This region is held close to the substrate when seeking a settlement
site.
In addition to the ventral mantle/gill bud, catecholamine-positive cells
have also been localised in the posterior-dorsal region (Figure
3c). This location matches that of a group of stereocilia (Figures
3b & d) that are associated with a shell feature known as the
posteriodorsal notch. The notch is a feature which leaves a visible
'track' in the shell during growth (Figures
3a & b), resulting in a small gap between the shell valves even
when they are closed (Figure
3a). As a result, this group of stereocilia will always have some
degree of contact with the water surrounding the animal.
| Figure 3a posteriodorsal notch | Figure 3b stereocilia under notch |
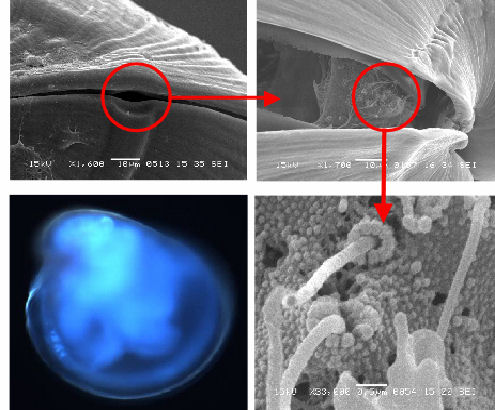 |
|
| Figure 3c Catecholamine signal (C) and notch region (N) | Figure 3d stereocilia under notch |
Serotonin
Like the catecholamines, serotonin has been implicated in the control
of ciliary activity of invertebrate larvae, but has also been identified
as having a more fluid role in other larval organisms. For example,
in the larvae of the sea star Patiriella regularis serotonin
has been identified in the ciliated feeding bands of the early larva,
suggesting a role in control of ciliary activity. However as the larvae
develop, serotonin redistributes within the larva, suggesting the activity
of serotonin is being redirected towards settlement (Chee and Byrne
1999).
Results from this study suggest a similar process may be occurring in
the larva of Crassostrea gigas. 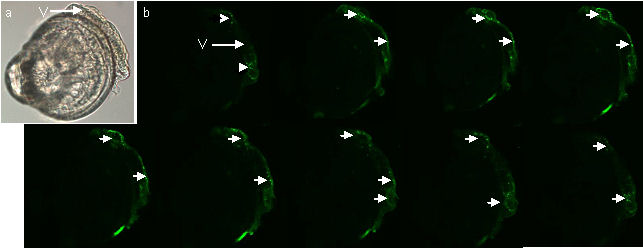
Figure 4 (a) LM image of a mid term veliger stage larva (b) Optical sections from CLSM showing serotonin sinal in the velum (arrows). Note that there is no signal from the rest of the larva.
Figures 4a & b above show a mid-term veliger stage
larva in which the antibody-labelled serotonin is restricted to the
velum (the swimming/feeding organ of this planktonic larva).
In contrast to the veliger, the late-stage pediveliger features serotonin
signals in the region of the mantle and gill bud (figure 5 & 6).
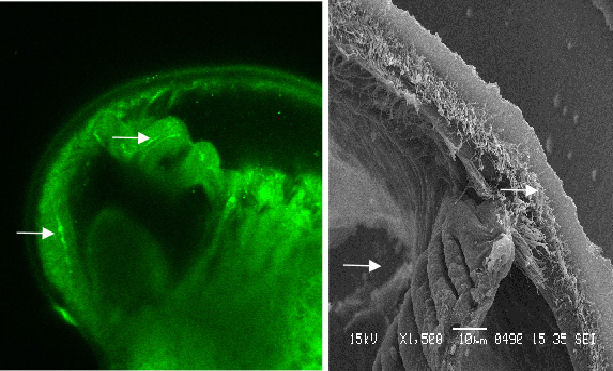 |
Figure 5 Serotonic image and contrasting SEM. Note that the signal corresponds with the gill bud and the beginnings of cilia tracts |
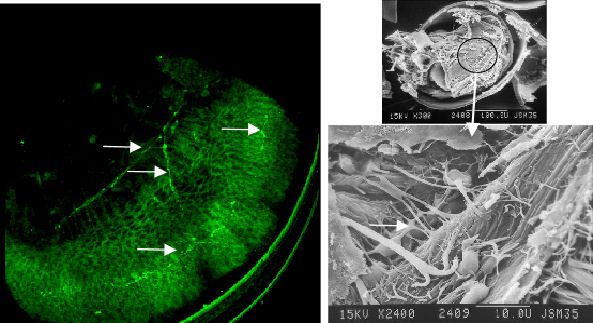 |
Fig. 6 Serotonin signal from the mantle musculature region of C.gigas pediveliger |
Additional CLSM work
In addition to the serotonin and catecholamine work this study also
aims to identify the cells bearing the large pre-oral cilia of the larval
velum. The nuclear stain Topro-3 is ideal for this work, enabling the
staining of the large nuclei of these cells. Initial results are encouraging
and suggest enumeration of nuclei of these cells is feasible (figure
7).
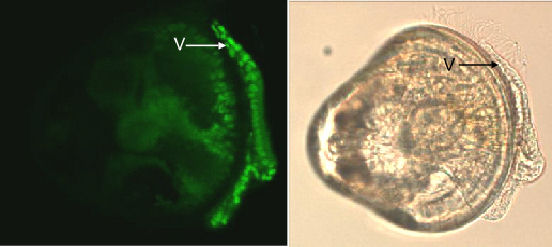 |
Figure 7. Topro-3 nuclei staining of the velum (V), and the corresponding LM image of the veliger. |
Summary
Initial results from this study suggest an association of both serotonin
and catecholamines with ciliated structures seen with SEM. Catecholamines
have been localised in the posteriodorsal region, an area associated
with stereocilia. Results suggest that serotonin signal in the veliger
is localised in the velum, with little or no signal being detected from
the mantle. Both catecholamines and serotonin have been identified in
the mantle and the gill bud of the pediveliger, specifically in those
areas bearing the significant and organised ciliation in the form of
cilia tracts. This is suggestive of the serotonin function changing
through development, altering from ciliary control of the feeding/swimming
cilia bands of the velum to a role within the mantle in settlement.
References
| Chee F, Byrne M (1999) Development of the larval serotonergic nervous system in the sea star Patiriella | |
| regularis as revealed by confocal imaging. Biological Bulletin 197: 123-131 | |
| Croll RP, Dickinson AJG (2004) Form and function of the larval nervous system in molluscs. Invertebrate | |
| Reproduction & Development 46: 173-187 | |
| Croll RP, Jackson DL, Voronezhskaya EE (1997) Catecholamine-containing cells in larval and postlarval | |
| bivalve molluscs. Biological Bulletin 193: 116-124 | |
Acknowledgements
I would like to thank the Malacological Society of London for the centenary
grant that has made this piece of work possible and for the conference
opportunities to present my research to a wider audience. Finally, Dr
Simon Cragg for his continued support and input, and Matthew Harris
for his assistance with my larval cultures.