

 RESEARCH
GRANT REPORT
RESEARCH
GRANT REPORT Josh Auld, Department of Biological Sciences, University of
Pittsburgh
Introduction and Background
Most organisms experience variation in biotic and abiotic environmental
factors during ontogeny. To persist, they must survive and reproduce
despite such environmental variability. Many organisms respond to
environmental variation with conditional strategies that involve phenotypic
changes based on environmental conditions (i.e. phenotypic plasticity).
An understanding of how environmental variation affects the interaction
between genotype and phenotype is fundamental to interpreting the
process of evolution in natural systems. Many studies have documented
how organisms respond to a single type of environmental variation,
but in nature many environmental factors can vary simultaneously.
Therefore, to understand the evolution of phenotypes in natural populations
we have to understand the context in which phenotypes are expressed
and how environmental factors interact to affect the expression and
evolution of the phenotype.
I am examining the effects and consequences of environmental variation
on phenotypes by examining two kinds of environmental factors that
interact to affect the phenotype: predation risk and mate availability.
These factors directly affect survival and reproduction and are therefore
closely tied to fitness, making them excellent factors for a study
of how interacting environmental factors affect a species' ecology
and evolution. An increase in predation risk leads to the expression
of inducible defenses that increase survival in the presence of a
predator. However, inducible defenses frequently incur a cost of slower
growth or decreased reproduction compared with the "no-predator"
phenotype. Variation in mate availability plays a crucial role in
determining mating systems and is a product of population size and
structure. Responding to variation in mate availability can lead to
patterns of inbreeding and outbreeding that deviate from random breeding.
In nature, organisms will experience variation in predation risk and
mate availability simultaneously.
Inducible defenses and mating systems are intimately linked because
inducible defenses frequently involve adjustment of traits such as
activity that also determine mating and reproductive success. Simultaneously,
changes in the mating system can lead to inbreeding or outbreeding
depression and therefore influence the development of an organism
and the production of an inducible defense. Responding to variation
in predation risk and mate availability frequently involves plastic
adjustment of the same traits (e.g. age or size at first reproduction).
Therefore, to understand the expression and evolution of defensive
and mating-system phenotypes in nature we must examine their interaction.
To explore how organisms respond to two independent sources of environmental
variation, I am examining the effects of predator-induced plasticity
on mating system expression and the implications of changes in the
mating system on the production of an inducible defense.
Proposed research
In my grant application, I proposed research to address how the effects
of plasticity in defensive and mating-system phenotypes interact in
the hermaphroditic freshwater snail Physa acuta.
Using a combination of laboratory and outdoor mesocosm experiments
to decompose these reciprocal effects, I have made progress towards
addressing two major hypotheses.
Hypothesis 1: Predator environments affect the mating system of
a prey population by increasing the proportion of selfed progeny produced.
Predators can reduce prey density by thinning the prey population
and induce changes in prey traits that affect the pool of available
mates. If mates are available and inbreeding depression is high, individuals
should mate and reproduce shortly after reaching sexual maturity.
However, if mates are not available, individuals should wait a set
amount of time before inbreeding to avoid the costs of inbreeding
depression (Tsitrone et al., 2003). Concurrently, size-selective
predation on small prey favours rapid growth and delayed reproduction
whereas predation on large individuals favours slow growth and early
reproduction (Stearns and Koella 1986). The combination of decreased
mate availability due to predator thinning and delayed maturity (which
should truncate the time delay before self-fertilization) induced
by predators may result in an increase in the proportion of progeny
produced by self-fertilization.
Hypothesis 2: Inbreeding impairs the production of prey defenses.
I expect that with subsequent inbreeding, individual growth rate will
decrease.
Thus, the development of morphological and life-history defensive
phenotypes will be retarded in inbred individuals relative to outbred
individuals due to their reduced growth potential. This decrease in
plasticity should increase with subsequent inbreeding.
Research to date
Predators induce changes in the life-history traits of snails that
likely affect the mating system. To examine how predators influence
the mating system by 1) inducing changes in the timing of reproduction
and 2) changing how individuals respond to mate availability, I experimentally
examined how snails alter their life histories under combinations
of predator presence and mate availability. I collected ten adult
snails from a single, high-density (outcrossed) population and placed
them in the lab for oviposition. Progeny from ten clutches were used
in a full-factorial experimental combination of two predator treatments
(no predator and caged crayfish) crossed with two mate-availability
treatments (isolation and mate available three times a week for three
hours at a time). These four treatments were replicated ten times
for a total of 40 experimental units (1-L plastic tubs with weekly
water changes and constant ad libitum food rations). I quantified
growth rate, morphological changes in shell shape, age / size at first
reproduction, progeny success (survival and growth rate), and fecundity.
In the absence of predators, snails that were isolated delayed reproduction
compared to snails with partners. However, in the presence of predators,
snails with and without mates delayed reproduction (Fig. 1). With
crayfish, there was no additional waiting time prior to self-fertilizing.
Thus, when predators are present, there is a reduced probability of
locating a mate prior to initiating reproduction. Hence, individuals
are more likely to self-fertilize prior to finding a mate in the presence
of a predator than in the absence of a predator. I am currently testing
this hypothesis further by using microsatellite allele frequency data
to evaluate how the genetic mating system changes in the presence
/ absence of predators.
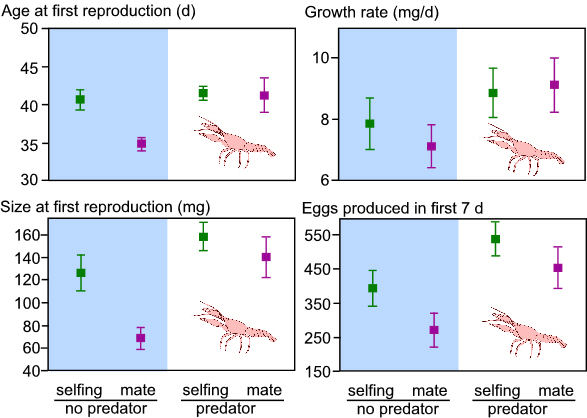 |
Figure 1. The effects predator presence and sexual partner availability on age and size at first reproduction, growth rate, and the number of eggs produced in the first week of reproduction. Data are means + 1 s.e.m. |
To evaluate the prediction that inbreeding impairs the production
of prey defenses, I am conducting the same experiment after several
generations of self-fertilization or outcrossing in the lab. I expect
that with subsequent inbreeding, growth rate and fecundity will decrease.
Also, I am measuring the same morphological and life-history traits
for inbred and outbred progeny to determine if inbreeding affects
the plasticity of snail traits.
It is important to be able to compare snails produced through self-fertilization
to an "outcrossed" control line of snails in order to control
for any inadvertent selection that occurs under lab conditions and
to control for any general environmental affects that differ between
generations. In my original attempt at this (August - December 2005),
I obtained snails for the second-generation experiment from my first-generation-experiment
snails. It was obvious that one generation of self-fertilization had
detrimental consequences on fitness (Fig. 2).
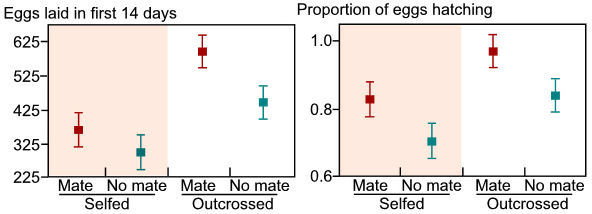 |
Figure 2. The effects of sexual partner availability and mating system on the number of eggs laid and the proportion of eggs hatching. These data are averaged across predator treatment. All data are means + 1 s.e.m |
Selfed snails laid 36% fewer eggs than outcrossed snails and 15% more of their eggs did not hatch. Thus, self-fertilization impairs fitness and also impairs the production of predator-induced shell morphology (data not shown). However, despite these obvious differences between the first-generation traits and the drastic reduction in fitness associated with self-fertilization, my outcrossing control lines were intermediate in many measures between the generation-1 snails and the generation-2 snails produced through self-fertilization. This suggests that my outcrossing treatments were occasionally self-fertilizing. To correct for this and obtain higher-quality results, I have implemented a new protocol for outcrossing snails. In this protocol, "outcrossing" lines are given consistent access to a mate (marked with red paint), which is changed everyday for one month. In this way, outcrossing parents potentially have access to 30 different mates from which they can obtain allosperm (individuals of this species can store sperm for ~3 months; Wethington and Dillon 1991). This new protocol is having the desired effect of more accurately mimicking an outcrossing-selfing comparison. I am currently completing the generation-1 experiment and approximately 1 month into the generation-2 experiment. This change in protocol will allow a better comparison.
Conclusions and Significance
Life persists because organisms survive and reproduce despite environmental
variation. To understand how organisms accomplish this, we need information
on how individuals integrate their phenotypes to simultaneously survive
predation, find mates, and maximize reproduction. This investigation
into the interactions between plasticity in defensive and mating-system
phenotypes will improve understanding of how inducible defenses are
expressed in nature (in the context of the mating system) and how
the mating system is affected by biotic interactions (such as predation
risk).
The funding from the Malacological Society of London has been integral
to the completion of this phase of the work, and I am extremely grateful
for the financial assistance.
Literature Cited
| Stearns, SC and JC Koella. 1986. The evolution of phenotypic plasticity in life-history traits: | |
| predictions of reaction norms for age and size at maturity. Evolution 40: 893-913. | |
| Tsitrone, A, S Duperron, and P David. 2003. Delayed selfing as an optimal mating strategy | |
| in preferentially outcrossing species: theoretical analysis of the optimal age at first reproduction in relation to mate availability. Am. Nat. 162: 318-331 | |
| Wethington, AR, and RT Dillon. 1991. Sperm storage and evidence for multiple insemination | |
| in a natural population of the freshwater snail, Physa. Am. Malacol. Bull. 9:99-102. | |